COMPARISON OF THE EFFECTIVENESS OF REMDESIVIR VERSUS FAVIPIRAVIR ON CLINICAL IMPROVEMENT AND MORTALITY AMONG PATIENTS WITH COVID-19 PNEUMONIA: A RETROSPECTIVE SINGLE-CENTER STUDY
DOI:
https://doi.org/10.55374/jseamed.v7.151Keywords:
COVID-19, Remdesivir, Favipiravir, Effectiveness, Mortality, Clinical improvementAbstract
Background: Antiviral drug administration in the early phase of COVID-19 during peak viremia can reduce the progression to severe disease. The optimal antiviral treatment against severe coronavirus disease 2019 (COVID‐19) has not been proven.
Objective: The study aimed to examine the effectiveness of remdesivir versus favipiravir to treat patients with COVID-19 pneumonia on clinical improvement and mortality.
Methods: This retrospective observational cohort study was conducted in the modular intensive care unit and cohort ward from 1 June 2021 to 31 December 2021. Patients were screened for COVID-19 pneumonia. A propensity score was used to handle selection bias and potential confounding factors. The propensity score estimation was obtained from the multivariable logistic regression model, including prognostic covariates. Then 1:1 matching was performed. Finally, the balance after matching was checked concerning the p-value.
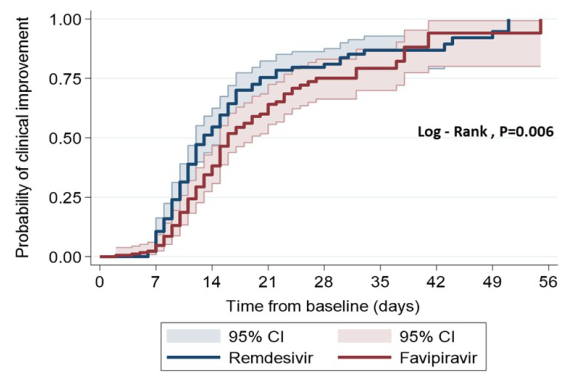
Results: Overall, 362 patients were matched using propensity score analysis; they were enrolled and divided in 2 groups: remdesivir and favipiravir (181:181). Remdesivir was associated with an increased proportion of clinical improvement (70.72 vs. 56.91%, adjusted HR=1.52 [1.16-2.01]; p=0.002), reduced inhospital mortality (adjusted HR=0.68 [0.47-0.99]; p=0.047), an increased proportion of being free from the use of a high flow nasal cannula (HFNC) and a low flow oxygen cannula (LFNC) (74.34 vs. 56.10%, adjusted HR 1.79 [1.32-2.45]; p<0.001; 86.4% vs. 74.8, adjusted HR=1.34 [1.01-1.78]; p=0.037, respectively), increased median survival time (26 vs. 24 days, median survival time difference of 2 days [IQR, 2-6]; p=0.048). In addition, patients treated with remdesivir showed a significantly higher proportion of discharge from the hospital measured using the WHO ordinary scale (66.85 vs. 53.04%, adjusted HR =1.19 [1.01-1.41]; p=0.035).
Conclusion: Among hospitalized patients with COVID-19 pneumonia, receiving oxygen supplementation, remdesivir was associated with increased clinical improvement, reduced in-hospital mortality and reduced need for HFNC and LFNC.
Downloads
Metrics
References
Sakunkoo P, Wangmook K, Maneemin N, Jirapornkul C, Koh D. Death from COVID-19 in Thailand 2020-2021. Epidemiol Int J 2022; 6: 000222. DOI: https://doi.org/10.23880/EIJ-16000222
Olender SA, Walunas TL, Martinez E, Perez KK, Castagna A, Wang S, et al. Remdesivir Versus Standard-of-Care for Severe Coronavirus Disease 2019 Infection: An Analysis of 28-Day Mortality. Open Forum Infect Dis 2021; 8: 1–9. DOI: https://doi.org/10.1093/ofid/ofab278
Joo EJ, Ko JH, Kim SE, Kang SJ, Baek JH, Heo EY, et al. Clinical and Virologic Effectiveness of Remdesivir Treatment for Severe Coronavirus Disease 2019 (COVID-19) in Korea: a Nationwide Multicenter Retrospective Cohort Study. J Korean Med Sci 2021; 36: 1–13. DOI: https://doi.org/10.3346/jkms.2021.36.e83
Frost MT, Jimenez-Solem E, Ankarfeldt MZ, Nyeland ME, Andreasen AH, Petersen TS. The Adaptive COVID-19 Treatment Trial-1 (ACTT-1) in a real-world population: a comparative observational study. Crit Care 2020; 24: 3–5. DOI: https://doi.org/10.1186/s13054-020-03406-3
Ansems K, Grundeis F, Dahms K, Mikolajewska A, Thieme V, Piechotta V, et al. Remdesivir for the treatment of COVID-19. Cochrane Database Syst Rev 2021; 8: CD014962. DOI: https://doi.org/10.1002/14651858.CD014962
Almoosa Z, Saad M, Qara S, Mustafa M, Mansour A, Alshab D, et al. Favipiravir versus standard of care in patients with severe COVID-19 infections: A retrospective comparative study. J Infect Public Health 2021; 14: 1247–53. DOI: https://doi.org/10.1016/j.jiph.2021.08.022
Cao B, Wang Y, Wen D, Liu W, Wang J, Fan G, et al. A Trial of Lopinavir–Ritonavir in Adults Hospitalized with Severe Covid-19. N Engl J Med 2020; 382: 1787–99. DOI: https://doi.org/10.1056/NEJMc2008043
Wang Y, Zhang D, Du G, Du R, Zhao J, Jin Y, et al. Remdesivir in adults with severe COVID-19: a randomised, double-blind, placebo-controlled, multicentre trial. Lancet 2020; 395: 1569–78. DOI: https://doi.org/10.1016/S0140-6736(20)31022-9
Ohl ME, Miller DR, Lund BC, Kobayashi T, Richardson Miell K, Beck BF, et al. Association of Remdesivir Treatment with Survival and Length of Hospital Stay among US Veterans Hospitalized with COVID-19. JAMA Netw Open 2021; 4: 1–14. DOI: https://doi.org/10.1001/jamanetworkopen.2021.14741
Department of Medical Services, Ministry of Public Health. Guidelines on clinical practice, diagnosis, treatment, and prevention of healthcare-associated infection for COVID-19 [Internet]. Nonthaburi; MOPH; 2021 [cited 2022Dec16].
Rubio-Rivas M, Mora-Luján JM, Formiga F, Arévalo-Cañas C, Lebrón Ramos JM, Villalba García MV, et al. WHO Ordinal Scale and Inflammation Risk Categories in COVID-19. Comparative Study of the Severity Scales. J Gen Intern Med 2022; 37: 1980–7. DOI: https://doi.org/10.1007/s11606-022-07511-7
Suwatanapongched T, Nitiwarangkul C, Sukkasem W, Phongkitkarun S. Rama CoRADS: Categorical Assessment Scheme of Chest Radiographic Findings for Diagnosing Pneumonia in Patients With Confirmed COVID-19. Ramathibodi Med J 2021; 44: 50–62. DOI: https://doi.org/10.33165/rmj.2021.44.2.251259
Gressens SB, Esnault V, De Castro N, Sellier P, Sene D, Chantelot L, et al. Remdesivir in combination with dexamethasone for patients hospitalized with COVID-19: A retrospective multicenter study. PLoS One 2022; 17: 7–13. DOI: https://doi.org/10.1371/journal.pone.0262564
Nguyen TL, Collins GS, Spence J, Daurès JP, Devereaux PJ, Landais P, et al. Doubleadjustment in propensity score matching analysis: choosing a threshold for considering residual imbalance. BMC Med Res Methodol 2017; 17: 1–8. DOI: https://doi.org/10.1186/s12874-017-0338-0
Agrawal U, Raju R, Udwadia ZF. Favipiravir: A new and emerging antiviral option in COVID-19. Med J Armed Forces India 2020; 76: 370–6. DOI: https://doi.org/10.1016/j.mjafi.2020.08.004
Beigel JH, Tomashek KM, Dodd LE, Mehta AK, Zingman BS, Kalil AC, et al. Remdesivir for the Treatment of Covid-19 — Final Report. N Engl J Med 2020; 383: 1813–26. DOI: https://doi.org/10.1056/NEJMoa2007764
Heil EL, Kottilil S. The Goldilocks Time for Remdesivir — Is Any Indication Just Right? N Engl J Med 2022; 386: 385–7. DOI: https://doi.org/10.1056/NEJMe2118579
Gottlieb RL, Vaca CE, Paredes R, Mera J, Webb BJ, Perez G, et al. Early Remdesivir to Prevent Progression to Severe Covid-19 in Outpatients. N Engl J Med 2022; 386: 305–15. DOI: https://doi.org/10.1056/NEJMoa2116846
Garibaldi BT, Wang K, Robinson ML, Zeger SL, Bandeen-Roche K, Wang MC, et al. Comparison of Time to Clinical Improvement with vs without Remdesivir Treatment in Hospitalized Patients with COVID-19. JAMA Netw Open 2021; 4: 1–14. DOI: https://doi.org/10.1001/jamanetworkopen.2021.3071
Benfield T, Bodilsen J, Brieghel C, Harboe ZB, Helleberg M, Holm C, et al. Improved Survival Among Hospitalized Patients With Coronavirus Disease 2019 (COVID-19) Treated With Remdesivir and Dexamethasone. A Nationwide PopulationBased Cohort Study. Clin Infect Dis. 2021;73: 2031–6. DOI: https://doi.org/10.1093/cid/ciab536
Chuah CH, Chow TS, Hor CP, Cheng JT, Ker HB, Lee HG, et al. Efficacy of Early Treatment With Favipiravir on Disease Progression Among High-Risk Patients With Coronavirus Disease 2019 ( COVID-19 ): A Randomized, Open-Label Clinical Trial Clin Infect Dis 2022; 75: e432-e439
Hassanipour S, Arab-Zozani M, Amani B, Heidarzad F, Fathalipour M, Martinezde-Hoyo R. The efficacy and safety of Favipiravir in treatment of COVID-19: a systematic review and meta-analysis of clinical trials. Sci Rep 2021; 11: 1–11. DOI: https://doi.org/10.1038/s41598-021-90551-6
Sheahan TP, Sims AC, Graham RL, Menachery VD, Gralinski LE, Case JB, et al. Broad-spectrum antiviral GS-5734 inhibits both epidemic and zoonotic coronaviruses. Sci Transl Med. 2017; 9: eaal3653. DOI: https://doi.org/10.1126/scitranslmed.aal3653
Szemiel AM, Merits A, Orton RJ, MacLean OA, Pinto RM, Wickenhagen A, et al. In vitro selection of Remdesivir resistance suggests evolutionary predictability of SARS-CoV-2. PLoS Pathog 2021; 17: 1–24. DOI: https://doi.org/10.1371/journal.ppat.1009929
Wang Y, Li P, Rajpoot S, Saqib U, Yu P, Li Y, et al. Comparative assessment of favipiravir and remdesivir against human coronavirus NL63 in molecular docking and cell culture models. Sci Rep 2021; 11: 1–13. DOI: https://doi.org/10.1038/s41598-021-02972-y
WHO Solidarity Trial Consortium. Remdesivir and three other drugs for hospitalised patients with covid-19: Final results of the who solidarity randomised trial and updated metaanalyses. The Lancet 2022; 399: 1941–53. DOI: https://doi.org/10.1016/S0140-6736(22)00519-0
Falcone M, Suardi LR, Tiseo G, Barbieri C, Giusti L, Galfo V, Forniti A, Caroselli C, Della Sala L, Tempini S, Okoye C, Monzani F, Menichetti F. Early Use of Remdesivir and Risk of Disease Progression in Hospitalized Patients With Mild to Moderate COVID-19. Clin Ther 2022; 44: 364-73. DOI: https://doi.org/10.1016/j.clinthera.2022.01.007
Kumar P, Kulkarni A, Sharma M, Nagaraja Rao P, Nageshwar Reddy D. Favipiravirinduced liver injury in patients with coronavirus disease 2019. J Clin Transl Hepatol 2021; 9: 276–8.
Kaur RJ, Charan J, Dutta S, Sharma P, Bhardwaj P, Sharma P, et al. Favipiravir use in COVID-19: Analysis of suspected adverse drug events reported in the WHO database. Infect Drug Resist 2020; 13: 4427–38. DOI: https://doi.org/10.2147/IDR.S287934
Leegwater E, Strik A, Wilms EB, Bosma LBE, Burger DM, Ottens TH, et al. Drug-induced Liver Injury in a Patient with Coronavirus Disease 2019: Potential Interaction of Remdesivir with P-Glycoprotein Inhibitors. Clin Infect Dis 2021; 72: 1256–8. DOI: https://doi.org/10.1093/cid/ciaa883
Downloads
Published
How to Cite
Issue
Section
License
The Journal of Southeast Asian Medical Research will hold the copyright to all published articles. The publisher's production department handles copyright forms once a manuscript is accepted and scheduled for publication.







