EFFECTS OF ULTRAVIOLET C (UVC) LIGHT AND DRY HEAT ON FILTRATION PERFORMANCE OF N95 RESPIRATOR MASK
DOI:
https://doi.org/10.55374/jseamed.v4i2.75Keywords:
COVID-19, N95 respirator mask, Ultraviolet C, Dry heatAbstract
Background: The emergence of the Coronavirus disease 2019 (COVID-19) pandemic caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV2) creates one of the most pressing issues with a severe shortage of personal protective equipment (PPE) particularly N95 respirators in healthcare settings worldwide. Recently, possible strategies to decontaminate disposable N95 respirators, including using ultraviolet C (UVC) irradiation and heat treatment, were reported to consider safely reusing the respirators. However, both methods create potential risks to reduce the ability of the respirator filter especially when exposed to these methods multiple times resulting in infectious agents passing through the filter.
Objective: The study aimed to ensure the effectiveness of UVC and dry heat to decontaminate N95 respirators.
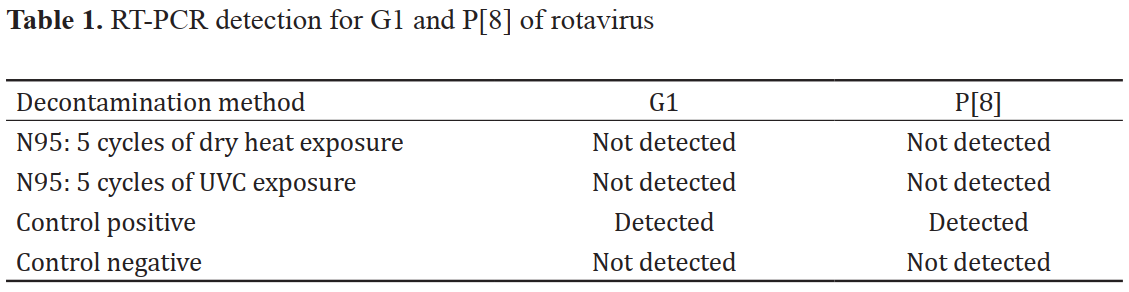
Methods: N95 respirators were exposed continually to UVC and dry heat at 70°C. Then the ability of the aerosol penetration was assessed by introducing an aerosol containing a rotavirus used as a delegate for SARS-CoV2. The existence of the rotavirus at both external (front) and internal surfaces (back) of the N95 respirators was investigated using RT-PCR.
Results: UVC and dry heat administered at a 30-minute cycle up to 5 cycles did not change the filtration performance of the N95 respirators. Our results suggested that the reuse of disposable N95 respirators decontaminated by either UVC or dry heat could be possible under the test conditions used.
Conclusion: To reuse N95 respirators, UVC and dry heat were useful to apply amid the pandemic of respiratory diseases.
Downloads
Metrics
References
Jernigan DB, Team CC-R. Update: Public health response to the coronavirus disease 2019 outbreak - United States, February 24, 2020. MMWR Morb Mortal Wkly Rep 2020; 69: 216-9. DOI: https://doi.org/10.15585/mmwr.mm6908e1
Cucinotta D, Vanelli M. WHO declares COVID-19 a pandemic. Acta Biomed 2020; 91: 157-60.
Gabutti G, d’Anchera E, Sandri F, Savio M, Stefanati A. Coronavirus: update related to the current outbreak of COVID-19. Infect Dis Ther 2020: 9: 1-13. DOI: https://doi.org/10.1007/s40121-020-00295-5
Liu J, Liao X, Qian S, Yuan J, Wang F, Liu Y, et al. Community transmission of severe acute respiratory syndrome coronavirus 2, Shenzhen, China, 2020. Emerg Infect Dis 2020; 26: 1320-3. DOI: https://doi.org/10.3201/eid2606.200239
Chan JF, Yuan S, Kok KH, To KK, Chu H, Yang J, et al. A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: a study of a family cluster. Lancet 2020; 395: 514-23. DOI: https://doi.org/10.1016/S0140-6736(20)30154-9
Morawska L, Cao J. Airborne transmission of SARS-CoV-2: The world should face the reality. Environ Int 2020; 139: 105730. DOI: https://doi.org/10.1016/j.envint.2020.105730
Siegel JD, Rhinehart E, Jackson M, Chiarello L, Committee HCICPA. 2007 Guideline for isolation precautions: preventing transmission of infectious agents in health care settings. Am J Infect Control 2007; 35(Suppl 2): S65- 164. DOI: https://doi.org/10.1016/j.ajic.2007.10.007
Janssen L, Ettinger H, Graham S, Shaffer R, Zhuang Z. The use of respirators to reduce inhalation of airborne biological agents. J Occup Environ Hyg 2013; 10: D97-D103. DOI: https://doi.org/10.1080/15459624.2013.799964
MacIntyre CR, Chughtai AA, Rahman B, Peng Y, Zhang Y, Seale H, et al. The efficacy of medical masks and respirators against respiratory infection in healthcare workers. Influenza Other Respir Viruses 2017; 11: 511-7. DOI: https://doi.org/10.1111/irv.12474
CDC P. Decontamination and reuse of filtering facepiece respirators. 2020.
Lindsley WG, Martin SB, Thewlis RE, Sarkisian K, Nwoko JO, Mead KR, et al. Effects of ultraviolet germicidal irradiation (UVGI) on N95 respirator filtration performance and structural integrity. J Occup Environ Hyg 2015; 12: 509-17. DOI: https://doi.org/10.1080/15459624.2015.1018518
Narla S, Lyons AB, Kohli I, Torres AE, Parks-Miller A, Ozog DM, et al. The importance of the minimum dosage necessary for UVC decontamination of N95 respirators during the COVID-19 pandemic. Photodermatol Photoimmunol Photomed 2020; 36:324-5. DOI: https://doi.org/10.1111/phpp.12562
Fischer RJ, Morris DH, van Doremalen N, Sarchette S, Matson MJ, Bushmaker T, et al. Effectiveness of N95 respirator decontamination and reuse against SARS-CoV-2 virus. Emerg Inf Dis 2020; 26:2253-5. DOI: https://doi.org/10.3201/eid2609.201524
Guntapong R, Tacharoenmuang R, Singchai P, Upachai S, Sutthiwarakom K, Komoto S, et al. Predominant prevalence of human rotaviruses with the G1P[8] and G8P[8] genotypes with a short RNA profile in 2013 and 2014 in Sukhothai and Phetchaboon provinces, Thailand. J Med Virol 2017; 89: 615-20. DOI: https://doi.org/10.1002/jmv.24669
Rengasamy S, King WP, Eimer BC, Shaffer RE. Filtration performance of NIOSHapproved N95 and P100 filtering facepiece respirators against 4 to 30 nanometer-size nanoparticles. J Occup Environ Hyg 2008; 5: 556-64. DOI: https://doi.org/10.1080/15459620802275387
Adhikari A, Mitra A, Rashidi A, Ekpo I, Schwartz J, Doehling J. Field evaluation of N95 filtering facepiece respirators on construction jobsites for protection against airborne ultrafine particles. Int J Environ Res Public Health 2018; 15: 1958. DOI: https://doi.org/10.3390/ijerph15091958
Perdiz D, Grof P, Mezzina M, Nikaido O, Moustacchi E, Sage E. Distribution and repair of bipyrimidine photoproducts in solar UV-irradiated mammalian cells. Possible role of Dewar photoproducts in solar mutagenesis. J Biol Chem 2000; 275: 26732-42. DOI: https://doi.org/10.1016/S0021-9258(19)61437-7
Fisher EM, Shaffer RE. A method to determine the available UV-C dose for the decontamination of filtering facepiece respirators. J Appl Microbiol 2011; 110: 287-95. DOI: https://doi.org/10.1111/j.1365-2672.2010.04881.x
Vo E, Rengasamy S, Shaffer R. Development of a test system to evaluate procedures for decontamination of respirators containing viral droplets. Appl Environ Microbiol 2009; 75: 7303-9. DOI: https://doi.org/10.1128/AEM.00799-09
Lore MB, Heimbuch BK, Brown TL, Wander JD, Hinrichs SH. Effectiveness of three decontamination treatments against influenza virus applied to filtering facepiece respirators. Ann Occup Hyg 2012; 56: 92-101.
Heimbuch BK, Wallace WH, Kinney K, Lumley AE, Wu CY, Woo MH, et al. A pandemic influenza preparedness study: use of energetic methods to decontaminate filtering facepiece respirators contaminated with H1N1 aerosols and droplets. Am J Infect Control 2011; 39: e1-9. DOI: https://doi.org/10.1016/j.ajic.2010.07.004
Rengasamy A, Zhuang Z, Berryann R. Respiratory protection against bioaerosols: literature review and research needs. Am J Infect Control 2004; 32: 345-54. 23. Chin AWH, Chu JTS, Perera MRA, Hui KPY, Yen HL, Chan MCW, et al. Stability of SARS-CoV-2 in different environmental conditions. The Lancet Microbe 2020; 1: e10. DOI: https://doi.org/10.1016/j.ajic.2004.04.199
Bernstein DI. Live attenuated human rotavirus vaccine, Rotarix. Semin Pediatr Infect Dis 2006; 17: 188-94. DOI: https://doi.org/10.1053/j.spid.2006.08.006
Kerdpanich A, Chokephaibulkit K, Watanaveeradej V, Vanprapar N, Simasathien S, Phavichitr N, et al. Immunogenicity of a live-attenuated human rotavirus RIX4414 vaccine with or without buffering agent. Hum Vaccin 2010; 6: 254-62. DOI: https://doi.org/10.4161/hv.6.3.10428
Pesavento JB, Crawford SE, Estes MK, Prasad BV. Rotavirus proteins: structure and assembly. Curr Top Microbiol Immunol 2006; 309: 189-219. DOI: https://doi.org/10.1007/3-540-30773-7_7
Downloads
Published
How to Cite
Issue
Section
License
The Journal of Southeast Asian Medical Research will hold the copyright to all published articles. The publisher's production department handles copyright forms once a manuscript is accepted and scheduled for publication.







