CORRELATION BETWEEN CLINICAL AND PATHOLOGIC FEATURES OF DIABETIC NEPHROPATHY
DOI:
https://doi.org/10.55374/jseamed.v6i0.113Keywords:
Diabetic nephropathy, Arteriolar hyalinosis, Interstitial fibrosis and tubular atrophy, Renal functionAbstract
Background: Diabetic nephropathy is the most common cause of end stage renal disease in Thailand. Renal biopsy remains the gold standard investigation to diagnose and classify diabetic nephropathy.
Objectives: In this study, we aimed to evaluate the correlation between clinical parameters and renal pathology classification among patients with type 2 diabetic and nephropathy.
Methods: We conducted an observational study and enrolled 63 patients undergoing renal biopsy between 1 January 2014 and 31 December 2018. Pathologic classification established by the Renal Pathology Society was used to assess the severity of histologic lesions in diabetic nephropathy. Clinical parameters including age, sex, duration, presence of diabetic retinopathy, blood urea nitrogen, creatinine, urine protein creatinine ratio, fasting plasma glucose and hemoglobin A1C were collected.
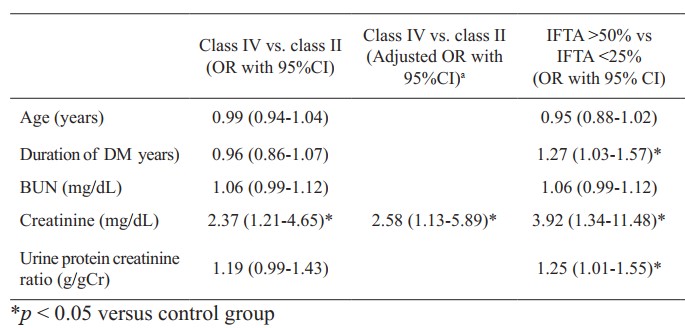
Results: At the time of biopsy, mean age was 50.25±11.46 years. Median duration of diabetes mellitus was 10 years with interquartile range (IQR) 3.75-12.00 years, mean serum creatinine was 2.44 ± 1.31 mg/dL and estimated glomerular filtration rate was 22.41±12.16 mL/min/1.73 m2. Based on the glomerular classification, 1 patient (1.6%) was in class I, 16 (25.3%) in class II, 25 (39.7%) in class III and 21 (33.3%) in class IV. Using multivariate analysis, class IV was associated with rising serum creatinine compared with class II [adjusted odds ratio (AOR)= 2.58; 95% CI= 1.13-5.89]. Patients with interstitial fibrosis and tubular atrophy (IFTA) <25%, 25-50% and >50% were observed in 10, 27 and 22 patients, respectively. Patients with IFTA >50% were significantly associated with duration of diabetes (OR=1.27; 95%CI=1.21-1.57), serum creatinine (OR=3.92; 95%CI=1.34-11.48) and urine protein (OR= 1.25; 95%CI=1.01-1.55) compared with patients with IFA<25%. Using multivariate analysis, only serum creatinine (AOR=3.48; 95%CI=1.23-12.65) was confirmed as independently correlated to IFTA >50% compared with IFTA <25%. A univariate analysis revealed no significant correlation between vascular indexes and renal function.
Conclusion: The results revealed that advanced glomerular lesions and high IFTA >50% correlated with impaired renal function in type 2 diabetic nephropathy
Downloads
Metrics
References
Nata N, Rangsin R, Supasyndh O, Satirapoj B. Impaired Glomerular Filtration Rate in Type 2 Diabetes Mellitus Subjects: A Nationwide Cross-Sectional Study in Thailand. J Diabetes Res 2020; 2020: 6353949. DOI: https://doi.org/10.1155/2020/6353949
Umanath K, Lewis JB. Update on Diabetic Nephropathy: Core Curriculum 2018. Am J Kidney Dis 2018; 71: 884-95. DOI: https://doi.org/10.1053/j.ajkd.2017.10.026
Satirapoj B, Adler SG. Comprehensive approach to diabetic nephropathy. Kidney Res Clin Pract 2014; 33: 121-31. DOI: https://doi.org/10.1016/j.krcp.2014.08.001
Mogensen CE, Christensen CK, Vittinghus E. The stages in diabetic renal disease. With emphasis on the stage of incipient diabetic nephropathy. Diabetes 1983; 32 (Suppl 2): 64-78. DOI: https://doi.org/10.2337/diab.32.2.S64
White KE, Bilous RW. Type 2 diabetic patients with nephropathy show structuralfunctional relationships that are similar to type 1 disease. J Am Soc Nephrol 2000; 11: 1667-73. DOI: https://doi.org/10.1681/ASN.V1191667
Tervaert TWC, Mooyaart AL, Amann K, Cohen AH, Cook HT, Drachenberg CB, et al. Pathologic classification of diabetic nephropathy. J Am Soc Nephrol 2010; 21: 556-63. DOI: https://doi.org/10.1681/ASN.2010010010
Oh SW, Kim S, Na KY, et al. Clinical implications of pathologic diagnosis and classification for diabetic nephropathy. Diabetes Res Clin Pract 2012; 97: 418-24. DOI: https://doi.org/10.1016/j.diabres.2012.03.016
Okada T, Nagao T, Matsumoto H, Nagaoka Y, Wada T, Nakao T. Histological predictors for renal prognosis in diabetic nephropathy in diabetes mellitus type 2 patients with overt proteinuria. Nephrology (Carlton) 2012; 17: 68-75. DOI: https://doi.org/10.1111/j.1440-1797.2011.01525.x
American Diabetes A. 2. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes-2021. Diabetes Care 2021; 44: S15-S33. DOI: https://doi.org/10.2337/dc21-S002
Sahay M, Mahankali RK, Ismal K, Vali PS, Sahay RK, Swarnalata G. Renal histology in diabetic nephropathy: A novel perspective. Indian J Nephrol 2014; 24: 226-31. DOI: https://doi.org/10.4103/0971-4065.132999
Suzuki Y, Ueno M, Hayashi H, Nishi S, Satou H, Karasawa R, et al. A light microscopic study of glomerulosclerosis in Japanese patients with noninsulin-dependent diabetes mellitus: the relationship between clinical and histological features. Clin Nephrol 1994; 42: 155-62.
Matsumae T, Jimi S, Uesugi N, Takebayashi S, Naito S. Clinical and morphometrical interrelationships in patients with overt nephropathy induced by non-insulin-dependent diabetes mellitus. A light- and electronmicroscopy study. Nephron 1999; 81: 41-8. DOI: https://doi.org/10.1159/000045244
Ruggenenti P, Gambara V, Perna A, Bertani T, Remuzzi G. The nephropathy of non-insulindependent diabetes: predictors of outcome relative to diverse patterns of renal injury. J Am Soc Nephrol 1998; 9: 2336-43. DOI: https://doi.org/10.1681/ASN.V9122336
Bohle A, Wehrmann M, Bogenschutz O, Batz C, Muller CA, Muller GA. The pathogenesis of chronic renal failure in diabetic nephropathy. Investigation of 488 cases of diabetic glomerulosclerosis. Pathol Res Pract 1991; 187: 251-9. DOI: https://doi.org/10.1016/S0344-0338(11)80780-6
Osterby R, Gall MA, Schmitz A, Nielsen FS, Nyberg G, Parving HH. Glomerular structure and function in proteinuric type 2 (non-insulin-dependent) diabetic patients. Diabetologia 1993; 36: 1064-70. DOI: https://doi.org/10.1007/BF02374500
Schwartz MM, Lewis EJ, Leonard-Martin T, Lewis JB, Batlle D. Renal pathology patterns in type II diabetes mellitus: relationship with retinopathy. The Collaborative Study Group. Nephrol Dial Transplant 1998; 13: 2547-52. DOI: https://doi.org/10.1093/ndt/13.10.2547
Okada T, Matsumoto H, Nakao T, Nagaoka Y, Yamada C, Shino T, et al. Relationship of renal lesions with urinary protein excretion in patients with overt diabetic nephropathy. Nihon Jinzo Gakkai Shi 1999; 41: 475-85.
n Y, Xu F, Le W, Ge Y, Zhou M, Chen H, et al. Renal histologic changes and the outcome in patients with diabetic nephropathy. Nephrol Dial Transplant 2015; 30: 257-66. DOI: https://doi.org/10.1093/ndt/gfu250
Taft JL, Nolan CJ, Yeung SP, Hewitson TD, Martin FI. Clinical and histological correlations of decline in renal function in diabetic patients with proteinuria. Diabetes 1994; 43: 1046-51. DOI: https://doi.org/10.2337/diabetes.43.8.1046
Ueno M, Kawashima S, Nishi S, Shimada H, Karasawa R, Suzuki Y, et al. Tubulointerstitial lesions in non-insulin dependent diabetes mellitus. Kidney Int Suppl 1997; 63: S191-4.
Satirapoj B. Tubulointerstitial Biomarkers for Diabetic Nephropathy. J Diabetes Res 2018; 2018: 2852398. DOI: https://doi.org/10.1155/2018/2852398
Afroz T, Sagar R, Reddy S, Gandhe S, Rajaram KG. Clinical and histological correlation of diabetic nephropathy. Saudi J Kidney Dis Transpl 2017; 28: 836-41.
Katakami N. Mechanism of Development of Atherosclerosis and Cardiovascular Disease in Diabetes Mellitus. J Atheroscler Thromb 2018; 25: 27-39. DOI: https://doi.org/10.5551/jat.RV17014
Poznyak A, Grechko AV, Poggio P, Myasoedova VA, Alfieri V, Orekhov AN. The Diabetes Mellitus-Atherosclerosis Connection: The Role of Lipid and Glucose Metabolism and Chronic Inflammation. Int J Mol Sci 2020; 21. DOI: https://doi.org/10.3390/ijms21051835
Dahl-Jorgensen K, Larsen JR, Hanssen KF. Atherosclerosis in childhood and adolescent type 1 diabetes: early disease, early treatment? Diabetologia 2005; 48: 1445-53. DOI: https://doi.org/10.1007/s00125-005-1832-1
Holman RR, Paul SK, Bethel MA, Matthews DR, Neil HA. 10-year follow-up of intensive glucose control in type 2 diabetes. N Engl J Med 2008; 359: 1577-89. DOI: https://doi.org/10.1056/NEJMoa0806470
Downloads
Published
How to Cite
Issue
Section
License
The Journal of Southeast Asian Medical Research will hold the copyright to all published articles. The publisher's production department handles copyright forms once a manuscript is accepted and scheduled for publication.







