PREVALENCE OF ABNORMAL GLUCOSE METABOLISM AMONG THAI OVERWEIGHT AND OBESE CHILDERN AND ADOLESCENTS
DOI:
https://doi.org/10.55374/jseamed.v3i2.41Keywords:
Impaired glucose tolerance, Imparied fasting glucose, Type 2 diabetes, Insulin resistanceAbstract
Background: The prevalence of childhood obesity is in creasingly worldwide. Abnormal glucose metabolism (ASM) including impaired fasting glucose (IFG), impaired glucose tolerance (IGT) and type 2 diabetes (T2DM) is a common endocrine complication among overweight and obese children and adolescents. Few studies of AGM are available in Thailand.
Objectives: The study aimed to establish the prevalence of AGM and identify associated complications among overweight and obese children and adolescents.
Methods: Data of overweight and obese children and adolescents, aged 6 to 18 years, undergoing oral glucose tolerance test (OGTT) at phramongkutklao Hospital were reviewed retrospectively. Fasting blood sugar, Hemoglobin Ale (HbA1c). fasting insulin, triglyceride, high-density lipoprotein, low-density lipoprotein, aspartate transaminase and alanine aminotransferase were derived from fasting measurement. Homeostatic model assessment was calculated to represent the insulin resistance.
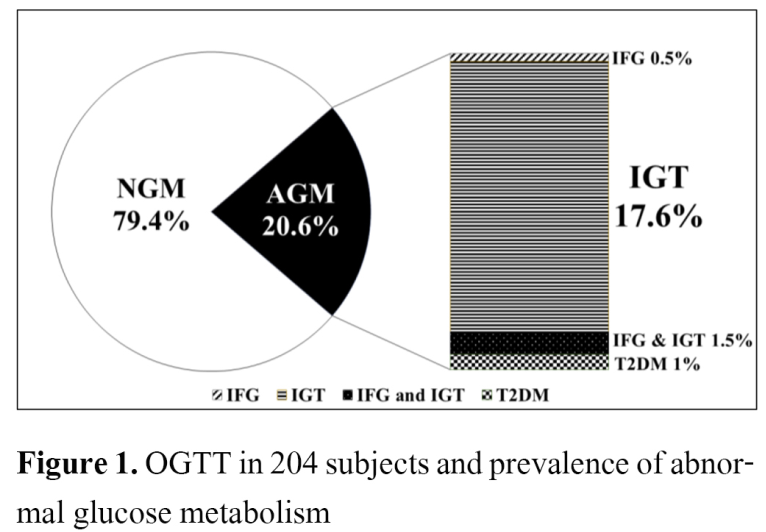
Result: A total of 204 children and adolescents (122 males) were included. Mean age was 12.1 ± 2.6 years and percent weight for height was 168.8 ± 25.4% The overall prevalence of AGM was 20.6%, 1 % had T2DM, 0.5% had IFG, 36% had IGT and 1.5% had combined IFG/IGT. Among the AGM, IGT was the most frequent subtype representing 92.8% (39/42 cases) but only 2 cases of T2DM were diagnosed in our study. The AGM group had significantly higher FPG (p=0.034), HbA1c (p=0.006) and cholesterol levels (p=0.043) than those of the nonabnormal glucose metabolism (NGM) group
Conclusion: Prevalences of AGM among overweight and obese children and adolescents were high. IGT was the most frequent group.
Downloads
Metrics
References
WHO [homepage on the Internet]. Geneva: WHO;c2016 [updated 2018 Feb 16; cited 2019 Mar 12]. Available from: https://www.who.int/en/news-room/fact-sheets/detail/obesity-and-overweight.
GBD 2015 Obesity Collaborators, Afshin A, Forouzanfar MH, Reitsma MB, Sur P, Estep K, Lee A, et al. Health Effects of Overweight and Obesity in 195 Countries over 25 Years. N Engl J Med 2017;377(1):13-27. DOI: https://doi.org/10.1056/NEJMoa1614362
Heymsfield SB, Wadden TA. Mechanism, Pathophysiology and management of obesity. N Engl J Med 2017;376:254-66. DOI: https://doi.org/10.1056/NEJMra1514009
Weiss R, Kaufman FR. Metabolic complications of childhood obesity. Diabetes Care 2008;31:S310-6. DOI: https://doi.org/10.2337/dc08-s273
Lee YS. Consequences of childhood obesity. Ann Acad Med Singapore 2009;38:75-81.
Weiss R, Taksali SE, Caprio S. Development of type 2 diabetes in children and adolescents. Current Diabetes Report 2006;6:182-7. DOI: https://doi.org/10.1007/s11892-006-0032-9
Tabak AG, Herder C, Rathmann W, Brunner EJ, Kivimaki M. Prediabetes: A high-risk state for diabetes development. Lancet 2012;379:2279-90. DOI: https://doi.org/10.1016/S0140-6736(12)60283-9
American Diabetes Association. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetesd 2019. Diabetes Care 2019;42(Suppl. 1):S13-S28. DOI: https://doi.org/10.2337/dc19-S002
American Diabetes Association. Children and Adolescents: Standards of Medical Care in Diabetes 2019.Diabetes Care. 2019;42(Suppl 1):S148-S164. DOI: https://doi.org/10.2337/dc19-S013
Weiss R, Taksali SE, Tamborlane WV, Burgert TS, Savoye M, Caprio S. Predictors of changes in oral glucose status in obese youth. Diabetes Care 2005;28:902-9. DOI: https://doi.org/10.2337/diacare.28.4.902
Anand SG, Mehta SD, Adams WG. Diabetes mellitus screening in pediatric primary care. Pediatrics 2006;118:1888-95. DOI: https://doi.org/10.1542/peds.2006-0121
Cambuli VM, Incani M, Pilia S, Congiu T, Cavallo MG, Cossu E, et, al. Oral glucose tolerance test in Italian overweight/obese children and adolescents results in a very high prevalence of impaired fasting glycaemia, but not of diabetes. Diabetes Metab Res Rev 2009;25(6):528-34. DOI: https://doi.org/10.1002/dmrr.980
Brufani C, Ciampalini P, Grossi A, Fiori R, Fintini D, Tozzi A, et, al. Glucose tolerance status in 510 children and adolescents attending an obesity clinic in Central Italy. Pediatr Diabetes 2010;11(1):47-54. DOI: https://doi.org/10.1111/j.1399-5448.2009.00527.x
Ruttanachuake S, Taweekul P, Aiumopas O, Suatvorawut O. Guideline for prevention and treatment in Thai obese children [Internet]. Thailand Pediatric Nutrition Association; 2014 [cited 2016 October 1]. Available from: http://www.thaipediatrics.org/Media/media-20161208151655.pdf.
The reference values of weight and height in Thai population age 1 day-19 years old. Department of Health. Ministry of Public Health. Thailand 1999.
Flynn JT, Kaelber DC, Baker-Smith CM, Blowey D, Carroll AE, Daniels SR, et, al. Clinical Practice Guideline for Screening and Management of High Blood Pressure in Children and Adolescents. Pediatrics 2017;140(3):e20171904. DOI: https://doi.org/10.1542/peds.2017-3035
Expert Panel on Integrated Guidelines for Cardiovascular Health and Risk Reduction in Children and Adolescents. Expert panel on integrated guidelines for cardiovascular health and risk reduction in children and adolescents: summary report. Pediatrics 2011;128 (suppl 5):S213–S256. DOI: https://doi.org/10.1542/peds.2009-2107C
Bamba V. Update on screening, etiology, and treatment of dyslipidemia in children. J Clin Endocrinol Metab 2014;99(9):3093-102. DOI: https://doi.org/10.1210/jc.2013-3860
Matthews DR, Hosker JR, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985;28:412-9. DOI: https://doi.org/10.1007/BF00280883
Jaruratanasirikul S, Thammaratchuchai S, Puwanant M, Mo-suwan L, Sriplung H. Progression from impaired glucose tolerance to type 2 diabetes in obese children and adolescents: a 3-6-year cohort study in southern Thailand. J Pediatr Endocrinol Metab 2016;29:1267-75. DOI: https://doi.org/10.1515/jpem-2016-0195
Shalitin S, Abrahami M, Lilos P, Phillip M. Insulin resistance and impaired glucose tolerance in obese children and adolescents referred to a tertiary-care center in Israel. Int J Obes (Lond) 2005;29(6):571-8. DOI: https://doi.org/10.1038/sj.ijo.0802919
Atabek ME, Pirgon O, Kurtoglu S. Assessment of abnormal glucose homeostasis and insulin resistance in Turkish obese children and adolescents. Diabetes Obes Metab 2007;9(3):304-10. DOI: https://doi.org/10.1111/j.1463-1326.2006.00601.x
Mohsin F, Mahbuba S, Begum T, Azad K, Nahar N. Prevalence of impaired glucose tolerance among children and adolescents with obesity. Mymensingh Med J 2012;21(4):684-90. DOI: https://doi.org/10.3329/birdem.v1i1.12382
Choudhary K, Mathur P, Garg M, Gupta PP. Prevalence of impaired glucose tolerance test and diabetes in overweight, obese and apparently healthy school going adolescents. Int J Contemp Pediatr 2017;4(3):1081-7. DOI: https://doi.org/10.18203/2349-3291.ijcp20171732
Sinha R, Fisch G, Teague B, Tamborlane WV, Banyas B, Allen K, et al. Prevalence of impaired glucose tolerance among children and adolescents with marked obesity. N Eng J Med 2002;346(11):802-10. DOI: https://doi.org/10.1056/NEJMoa012578
Maffeis C, Pinelli L, Brambilla P, Banzato C, Valzolgher L, Ulmi D, et, al. Fasting plasma glucose (FPG) and the risk of impaired glucose tolerance in obese children and adolescents. Obesity (Silver Spring) 2010;18(7):1437-42. DOI: https://doi.org/10.1038/oby.2009.355
Bonito PD, Pacifico L, Chiesa C, Valerio G, Del Giudice EM, Maffeis C. Impaired fasting glucose and impaired glucose tolerance in children and adolescents with overweight/ obesity. J Endocrinol Invest 2017;40(4):409-16. DOI: https://doi.org/10.1007/s40618-016-0576-8
Ek AE, Rossner SM, Hagman E, Marcus C. High prevalence of prediabetes in a Swedish cohort of severely obese children. Pediatric Diabetes 2015;16:117-28. DOI: https://doi.org/10.1111/pedi.12136
Panamonta O, Thamsiri N, Panamonta M. Prevalence of type II diabetes and metabolic syndrome among overweight school children in Khon Kaen, Thailand. J Med Assoc Thai 2010;93:56-60.
Weiss R, Bremer AA, Lustig RH. What is metabolic syndrome, and why are children getting it?. Ann N Y Acad Sci 2013;1281:123-40. DOI: https://doi.org/10.1111/nyas.12030
Bussler S, Penke M, Flemming G, Elhassan YS, Kratzsch J, Sergeyev E. Novel insights in the metabolic syndrome in childhood and adolescence. Horm Res Peadiate 2017;88:188-93. DOI: https://doi.org/10.1159/000479510
Ighbaliya A, Weiss R. Insulin Resistance, Prediabetes, Metabolic Syndrome: What Should Every Pediatrician Know?. J Clin Res Pediatr Endocrinol 2017;9(Suppl 2):49-57. DOI: https://doi.org/10.4274/jcrpe.2017.S005
Moran A, Jacobs DR, Steinberger J, Hong CP, Prineas R, Luepker, et al. Insulin resistance during puberty. Results from clamp studies in 357 children. Diabetes 1999;48(10):2039-44. DOI: https://doi.org/10.2337/diabetes.48.10.2039
Groot CJ, Grond JV, Delgado Y, Rings EH, Hannema SE, van den Akker EL. High predictability of impaired glucose tolerance by combining cardiometabolic screening parameters in obese children. J Pediatr Endocrinol Metab 2017;30(2):189-96. DOI: https://doi.org/10.1515/jpem-2016-0289
Ehehalt S, Wiegand S, Körner A, Schweizer R, Liesenkötter KP, Partsch CJ, et, al. Low association between fasting and OGTT stimulated glucose levels with HbA1c in overweight children and adolescents. Pediatr Diabetes 2017;18(8):734-41. DOI: https://doi.org/10.1111/pedi.12461
Wiegand S, Maikowski U, Blankenstein O, Biebermann H, Tarnow P, Grüters A. Type 2 diabetes and impaired glucose tolerance in European children and adolescents with obesity--a problem that is no longer restricted to minority groups. Eur J Endocrinol 2004;151(2):199-206. DOI: https://doi.org/10.1530/eje.0.1510199
Downloads
Published
How to Cite
Issue
Section
License
The Journal of Southeast Asian Medical Research will hold the copyright to all published articles. The publisher's production department handles copyright forms once a manuscript is accepted and scheduled for publication.







